Rene Becker, Tessel Bouwens, Esther C. F. Schippers, Toon van Gelderen, Michiel Hilbers, Sander Woutersen, Joost N. H. Reek
Chemistry – A European Journal, 2019, 25(61), 13921-13929
DOI: 10.1002/chem.201902514
Abstract
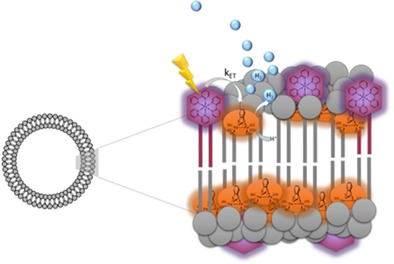
Artificial photosynthesis—the direct photochemical generation of hydrogen from water—is a promising but scientifically challenging future technology. Because nature employs membranes for photodriven reactions, the aim of this work is to elucidate the effect of membranes on artificial photocatalysis. To do so, a combination of electrochemistry, photocatalysis, and time‐resolved spectroscopy on vesicle‐embedded [FeFe]hydrogenase mimics, driven by a ruthenium tris‐2,2′‐bipyridine photosensitizer, is reported. The membrane effects encountered can be summarized as follows: the presence of vesicles steers the reactivity of the [FeFe]‐benzodithiolate catalyst towards disproportionation, instead of protonation, due to membrane characteristics, such as providing a constant local effective pH, and concentrating and organizing species inside the membrane. The maximum turnover number is limited by photodegradation of the resting state in the catalytic cycle. Understanding these fundamental productive and destructive pathways in complex photochemical systems allows progress towards the development of efficient artificial leaves.