Wenlin Zhang, Eva J. Meeus, Lei Wang, Lu-Hua Zhang, Shuangcheng Yang, Bas de Bruin, Joost N. H. Reek, Fengshou Yu
ChemSusChem, 2022, 15(3), e202102379
DOI: 10.1002/cssc.202102379
Abstract
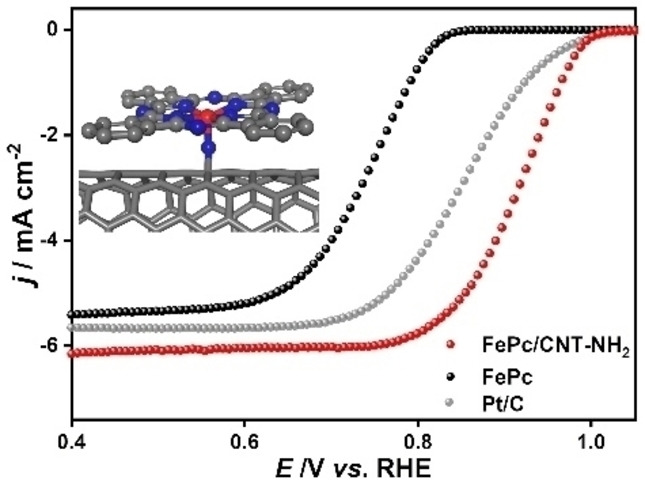
Interaction of the spheres: Molecular iron phthalocyanine is supported on functionalized multi-walled carbon nanotubes through axial coordination interaction and π-π stacking. The formed axial coordination configuration N−Fe−N4 can effectively break the planar structure of Fe−N4 and enhance the oxygen reduction performance with a half-wave potential of E1/2=0.92 V, which far exceeds commercial Pt/C with 0.85 V.
Precise regulation of the electronic states of catalytic sites through molecular engineering is highly desired to boost catalytic performance. Herein, a facile strategy was developed to synthesize efficient oxygen reduction reaction (ORR) catalysts, based on mononuclear iron phthalocyanine supported on commercially available multi-walled carbon nanotubes that contain electron-donating functional groups (FePc/CNT-R, with “R” being −NH2, −OH, or −COOH). These functional groups acted as axial ligands that coordinated to the Fe site, confirmed by X-ray photoelectron spectroscopy and synchrotron-radiation-based X-ray absorption fine structure. Experimental results showed that FePc/CNT-NH2, with the most electron-donating −NH2 axial ligand, exhibited the highest ORR activity with a positive onset potential (Eonset=1.0 V vs. reversible hydrogen electrode) and half-wave potential (E1/2=0.92 V). This was better than the state-of-the-art Pt/C catalyst (Eonset=1.00 V and E1/2=0.85 V) under the same conditions. Overall, the functionalized FePc/CNT-R assemblies showed enhanced ORR performance in comparison to the non-functionalized FePc/CNT assembly. The origin of this behavior was investigated using density functional theory calculations, which demonstrated that the coordination of electron-donating groups to FePc facilitated the adsorption and activation of oxygen. This study not only demonstrates a series of advanced ORR electrocatalysts, but also introduces a feasible strategy for the rational design of highly active electrocatalysts for other proton-coupled electron transfer reactions.