Thomas Freese, Jelmer T. Meijer, Maria B. Brands, Georgios Alachouzos, Marc C. A. Stuart, Rafael Tarozo, Dominic Gerlach, Joost Smits, Petra Rudolf, Joost N. H. Reek and Ben L. Feringa
EES Catalysis, 2024, 2(1), 262-275
DOI: 10.1039/d3ey00256j
Abstract
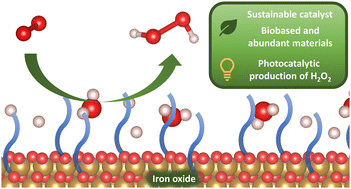
Hydrogen peroxide (H2O2) is a valuable green oxidant with a wide range of applications. Furthermore, it is recognized as a possible future energy carrier achieving safe operation, storage and transportation. The photochemical production of H2O2 serves as a promising alternative to the waste- and energy-intensive anthraquinone process. Following the 12 principles of Green Chemistry, we demonstrate a facile and general approach to sustainable catalyst development utilizing earth-abundant iron and biobased sources only. We developed several iron oxide (FeOx) nanoparticles (NPs) for successful photochemical oxygen reduction to H2O2 under visible light illumination (445 nm). Achieving a selectivity for H2O2 of >99%, the catalyst material could be recycled for up to four consecutive rounds. An apparent quantum yield (AQY) of 0.11% was achieved for the photochemical oxygen reduction to H2O2with visible light (445 nm) at ambient temperatures and pressures (9.4–14.8 mmol g−1 L−1). Reaching productivities of H2O2 of at least 1.7 ± 0.3 mmol g−1 L−1 h−1, production of H2O2 was further possible via sunlight irradiation and in seawater. Finally, a detailed mechanism has been proposed on the basis of experimental investigation of the catalyst’s properties and computational results.