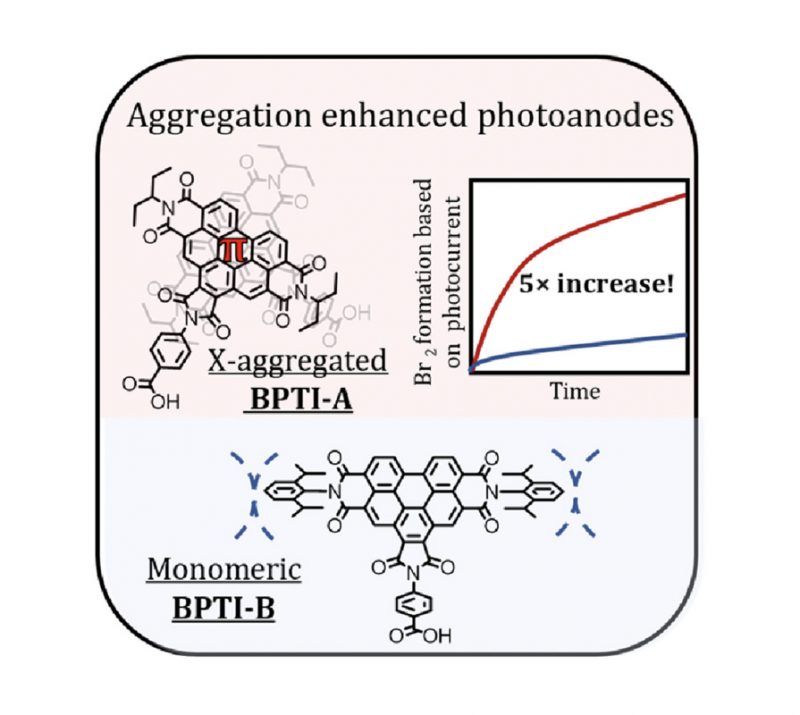
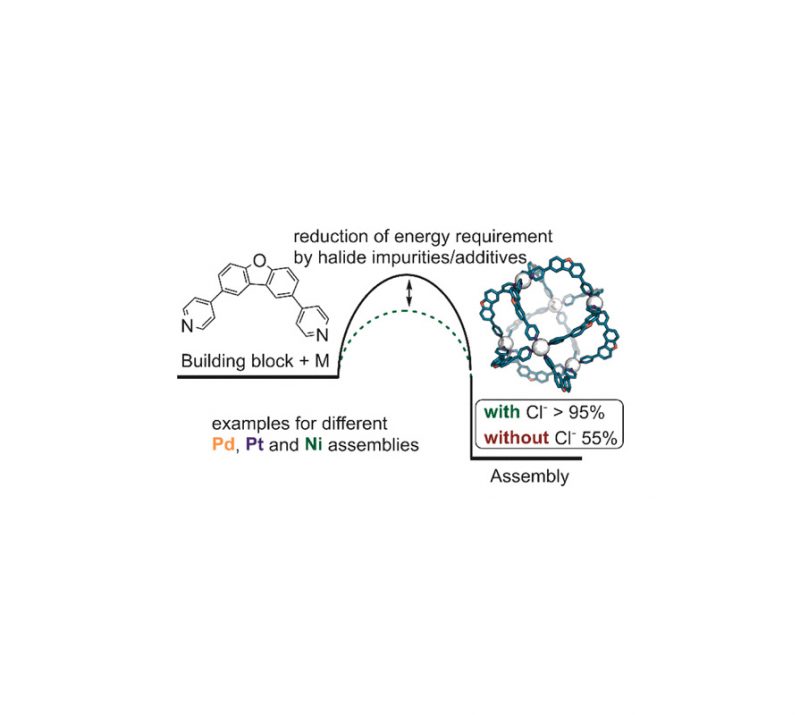
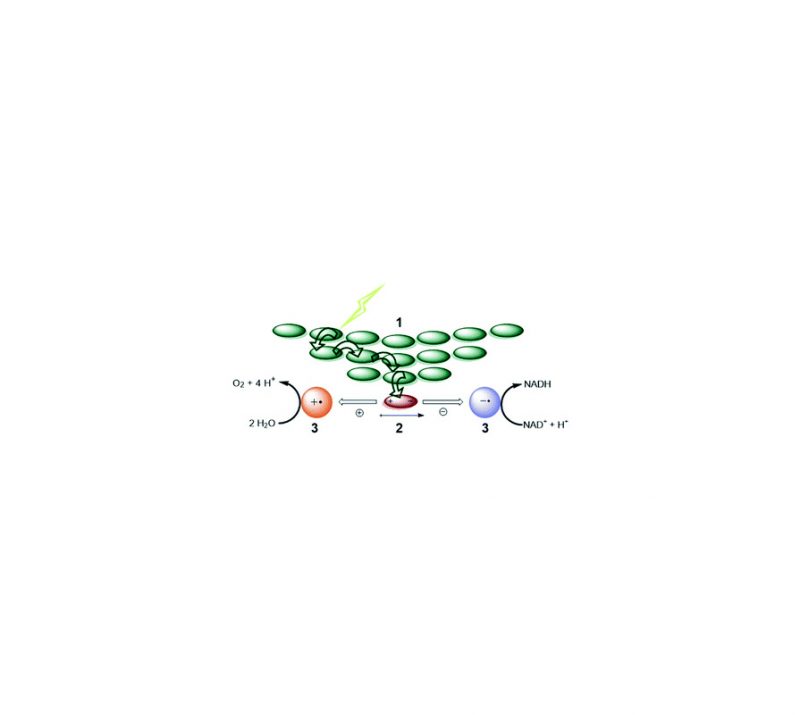
Sandra S. Nurttila, Riccardo Zaffaroni, Simon Mathew, Joost N. H. Reek
Chem. Commun., 2019, 55, 3081-3084
DOI: 10.1039/c9cc00901A
Abstract
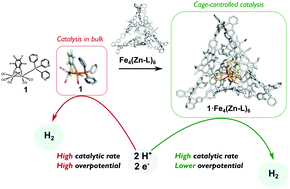
Hydrogen as a renewable fuel is viable when produced sustainably viaproton reduction catalysis (PRC). Many homogeneous electrocatalysts perform PRC with high rates, but they all require a large overpotential to drive the reaction. Natural hydrogenase enzymes achieve reversible PRC with potentials close to the thermodynamic equilibrium through confinement of the active site in a well-defined protein pocket. Inspired by nature, we report a strategy that relies on the selective encapsulation of a synthetic hydrogenase mimic in a novel supramolecular cage. Catalyst confinement decreases the PRC overpotential by 150 mV, and is proposed to originate from the cationic cage stabilizing anionic reaction intermediates within the catalytic cycle.