Felix J. de Zwart, Petrus C. M. Laan, Nicole S. van Leeuwen, Eduard O. Bobylev, Erika R. Amstalden van Hove, Simon Mathew, Ning Yan, Jitte Flapper, Keimpe J. van den Berg, Joost N. H. Reek, and Bas de Bruin
Macromolecules, 2022, 55, 21, 9690–9696
DOI: 10.1021/acs.macromol.2c01457
Abstract
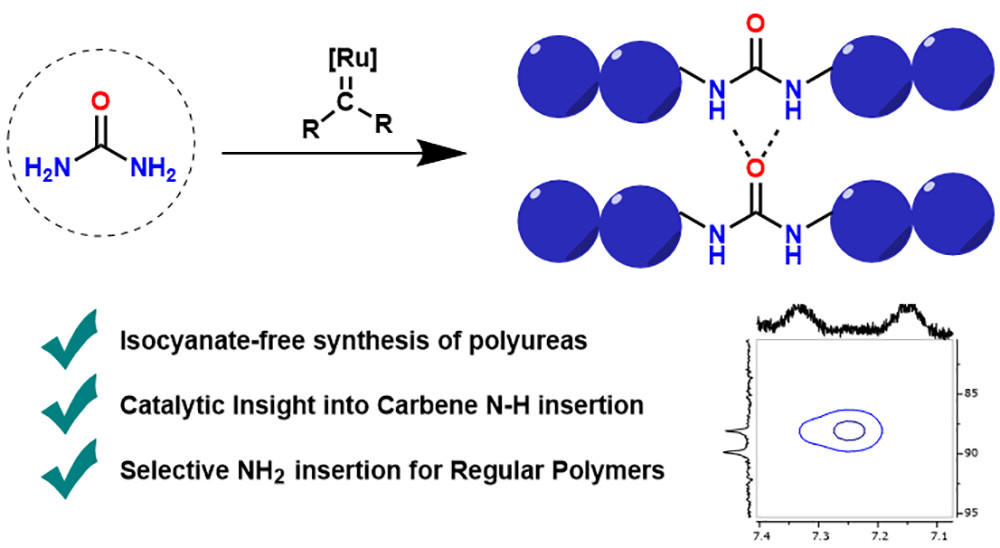
Polyureas have widespread applications due to their unique material properties. Because of the toxicity of isocyanates, sustainable isocyanate-free routes to prepare polyureas are a field of active research. Current routes to isocyanate-free polyureas focus on constructing the urea moiety in the final polymerizing step. In this study we present a new isocyanate-free method to produce polyureas by Ru-catalyzed carbene insertion into the N–H bonds of urea itself in combination with a series of bis-diazo compounds as carbene precursors. The mechanism was investigated by kinetics and DFT studies, revealing the rate-determining step to be nucleophilic attack on a Ru–carbene moiety by urea. This study paves the way to use transition-metal-catalyzed reactions in alternative routes to polyureas.